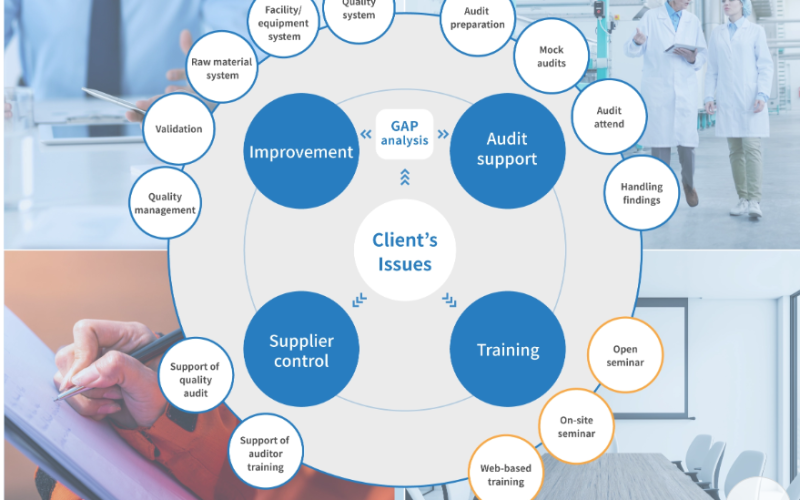
With extensive experience across diverse areas of the healthcare and life sciences industry, our team has successfully delivered numerous projects, providing compliant, effective, and efficient solutions. Clients can rely on QUALYinC for trusted expertise and sustained support to achieve and maintain GxP compliance across their operations. Whether support is required in Good Manufacturing Practice (GMP), Good Clinical Practice (GCP), Good Laboratory Practice (GLP), Digital Compliance, or other GxP standards, QUALYinC connects clients with subject-matter experts tailored to their specific needs.
_1773840546.png)